The Evolution and Impact of Single-Use Systems in Biopharma Manufacturing
The widespread adoption of single-use systems (SUS) has redefined biopharmaceutical manufacturing, offering greater operational flexibility, improved sterility assurance, and reduced capital investment. In this article, Ashutosh Mahamuni explores the transformative impact of SUS through a practitioner’s lens—drawing from real-world insights to examine their evolution, practical advantages, regulatory considerations, and long-term sustainability. As the industry shifts toward modular and data-driven production models, this piece highlights the critical role SUS will play in shaping the next generation of pharmaceutical innovation.
1. Introduction
- Historically, biopharmaceutical manufacturing relied on traditional stainless-steel bioreactors and equipment, which presented challenges related to cost, extensive cleaning validation, and contamination risks.
- The advent of SUS introduced disposable components, streamlining production workflows and ensuring compliance with stringent regulatory requirements.
- This shift has provided increased flexibility, particularly in the development and production of biologics and personalized medicine, enabling faster turnaround times and cost-efficient manufacturing.
2. Literature Review: The Rise of Single-Use Systems (2017–2019)
- Between 2017 and 2019, significant strides were made in the adoption of SUS, driven by the need for more flexible and cost-effective manufacturing processes. According to a 2018 report by BioProcess International, the global market for single-use bioprocessing was projected to grow at a CAGR of 12.8%, reflecting increasing industry reliance on disposable systems.
- For example, Amgen reported a 40% reduction in production downtime and a significant decrease in contamination incidents after transitioning to SUS. Similarly, Genentech streamlined its biologics production by implementing SUS, cutting cleaning validation times by 50% and reducing resource consumption.
3. Technical Analysis: Comparing Traditional Manufacturing vs. Single-Use Systems
| Feature | Traditional Stainless-Steel Systems | Single-Use Systems (SUS) |
|
High initial cost for equipment and infrastructure | Lower initial investment due to disposable components |
|
Requires extensive cleaning validation, sterilization, and downtime | Eliminates cleaning, reducing contamination risks and downtime |
|
Limited flexibility, requires fixed infrastructure | Highly adaptable for different production needs |
|
Expansion requires significant investment and time | Easier and faster to scale up or modify processes |
|
Higher risk due to cleaning effectiveness variability | Lower risk due to single-use, pre-sterilized components |
|
Requires large amounts of water and cleaning chemicals | Generates plastic waste, requiring advancements in sustainability |
- Reduced Contamination Risk: By eliminating the need for extensive cleaning validation, SUS significantly lowers the risk of microbial and cross-contamination. Cleaning validation is a critical process in traditional manufacturing where equipment must be thoroughly cleaned and tested before switching between different products to prevent cross-contamination.
- Cost-Effectiveness: Compared to traditional stainless-steel setups, SUS requires a lower initial capital investment and eliminates the need for costly cleaning and sterilization procedures. SUS implementation reduces capital investment and operational costs by eliminating the need for expensive stainless-steel infrastructure and associated cleaning expenses. For instance, the production of monoclonal antibodies (mAbs) has benefited significantly from SUS adoption. Companies like Lonza and WuXi Biologics have reported enhanced efficiency and cost reductions by integrating single-use bioreactors into their mAb manufacturing pipelines. This shift has enabled faster turnaround times, reduced labor costs, and improved scalability, making SUS a preferred choice for biologics production.
- Operational Flexibility: Single-use components enable rapid batch changeovers, increased scalability, and adaptability for various production demands. The modular nature of SUS allows for rapid process adjustments, making it ideal for adaptive manufacturing and small-batch production. The rise of SUS also created new opportunities for companies specializing in disposable bioprocessing equipment. Early adopters like Sartorius, Pall Corporation, and Thermo Fisher Scientific successfully capitalized on the shift by developing innovative single-use bioreactors, filtration units, and other components.
- Sustainability Considerations: While SUS reduces water and energy consumption, challenges remain regarding the environmental impact of disposable plastic components. Research efforts into biodegradable single-use plastics are gaining traction. Institutions like Virginia Tech are developing bioplastics from food waste, while companies like Constructive Bio are using microbial bio factories to engineer biodegradable plastics. Startups such a Sway are focusing on seaweed-based bioplastics, which offer compostable alternatives.
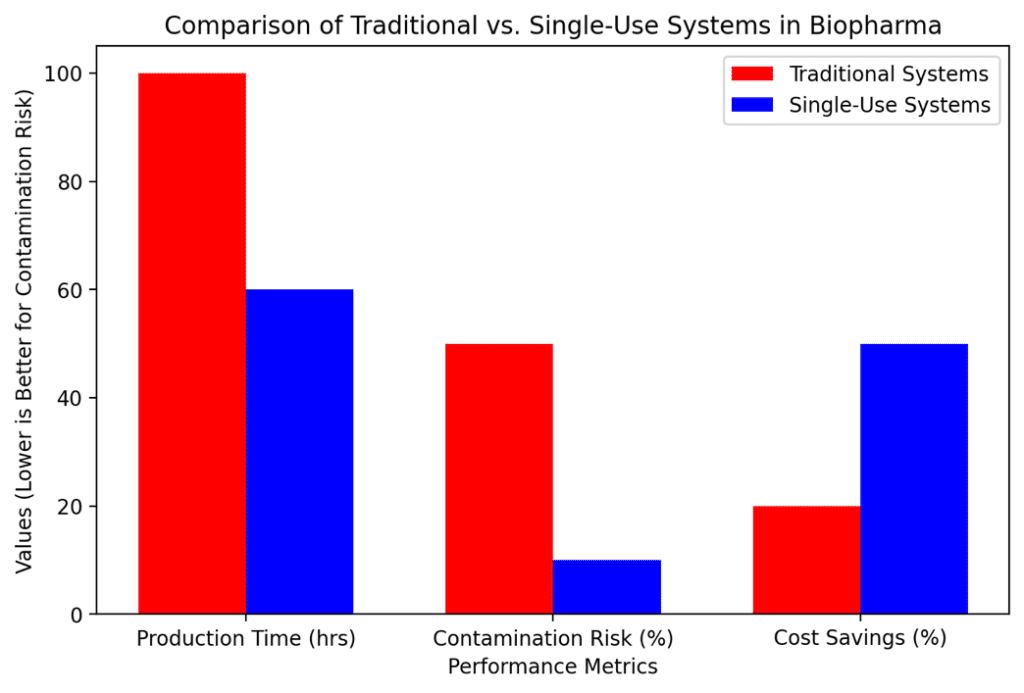
Figure 1: Comparative analysis of process efficiency between SUS and traditional stainless-steel systems.
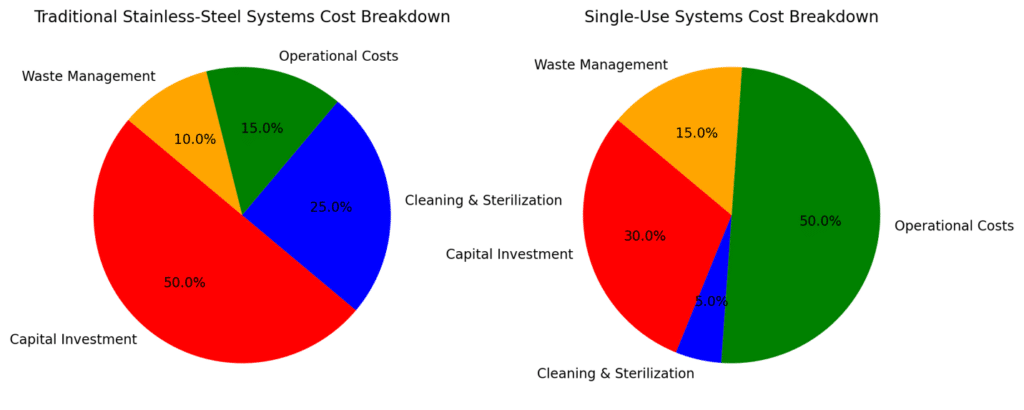
Figure 2: Overall, the transition to SUS can significantly reduce capital investment and cleaning costs, making it an attractive option for modern biopharma manufacturing. However, it comes with increased operational costs due to the recurring need for consumables, highlighting a key trade-off in adopting disposable bioprocessing solutions.
Single-use systems (SUS) are reshaping biopharmaceutical manufacturing by offering enhanced flexibility, faster turnaround, and reduced contamination risk. However, challenges persist—ranging from supply chain vulnerabilities and material compatibility to stringent regulatory scrutiny and environmental concerns. As demand outpaces supply, the industry must invest in sustainable materials, rigorous validation, and diversified sourcing. With automation and real-time monitoring driving efficiency, SUS is expanding beyond biologics into vaccines and gene therapies.
4. Conclusion
As biopharma continues to evolve, embracing the full potential of SUS—especially in personalized medicine and pandemic response—will be crucial to global healthcare innovation. To ensure continued adoption, the industry must address regulatory and environmental challenges. The future lies in advancing automation, sustainable materials, and broader application of SUS technologies.
References
- BioProcess International. (2018). Single-use bioprocessing market trends. BioProcess International Journal, 16(4), 45-52.
- FDA. (2017). Guidance on modernizing pharmaceutical manufacturing. U.S. Food and Drug Administration.
- EMA. (2018). Risk-based implementation of single-use technologies. European Medicines Agency.
- Amgen. (2019). Case study on SUS adoption: Efficiency and contamination reduction. Amgen Reports, 23(2), 58-65.
Author’s Bio:
Ashutosh Mahamuni is a graduate student in manufacturing optimization and continuous processing. His research on Spherofast, an innovative continuous pellet manufacturing technology, fostered a strong interest in single-use systems (SUS) as scalable and sustainable solutions in modern bioprocessing. He is dedicated to driving innovation in biopharma manufacturing. Check him out at: LinkedIn profile.